Oral dysfunction as a cause of malocclusion Linda D'Onofrio Oregon Health and Sciences University School of Dentistry, Portland, Oregon https://doi.org/10.1111/ocr.12277 INTRODUCTION
Most infants are beautiful because most children are born with normal
craniofacial shape, normal jaw relationship and potential for optimal airway. In
most newborn faces, the alveolar process easily accommodates the tongue and all
future teeth.
Nevertheless, orthodontists see multitudes of children
with abnormal jaw relationship, steep mandibular angle (SMA), anterior open
bite (AOB), high narrow palate (HNP), posterior cross bite (PCB) and suboptimal
facial development. While orthodontic referrals may begin at age seven, the
facial dysmorphology is often evident years earlier. When oral dysfunction goes
untreated, orofacial myofunctional disorder (OMD) can result. | 43
Orofacial myofunctional disorder includes dysfunction of
the lips, jaw, tongue and/or oropharynx that interferes with normal growth,
development or function of other oral structures, the
consequence of a sequence of events or
lack of intervention at critical periods, that result in malocclusion and
suboptimal facial development.
Oral
dysfunction can begin with our very first breath and with our very first
feeding.1 OMD can become apparent as children learn to speak2
and transition to table food.3 Most children with OMD are diagnosed
after experiencing articulation disorder, sleep- disordered breathing (SDB)4
or malocclusion.5 Orthodontic relapse, obstructive sleep apnoea
(OSA) and temporomandibular disorder6 are predictable consequences
of long- term oral dysfunction and OMD.
This
manuscript provides a brief narrative survey of ten areas of oral function
related to occlusal and facial development: breastfeeding, airway obstruction,
soft tissue restriction, mouth breathing, oral resting posture, oral habits,
swallowing, chewing, OMD over time and maternal oral dysfunction on the
developing foetus.
1
| IMPACT
OF BREASTFEEDING
Breastfeeding is the first and
perhaps most critical experience to facial development. Unlike with bottle
feeding, infants draw the breast deep into the mouth and the breast expands and
shapes the hard palate through repeated pressure and peristaltic wave.7
Breastfeeding requires jaw compression, which helps develop better masseter
muscles than does bottle feeding.8
Children exclusively breastfed appeared to have a
lower incidence of malocclusion later in life when compared to bottle- fed
babies. Studies demonstrated exclusive breastfeeding had an inverse correlation
to AOB,9 PCB, overjet and other malocclusions.10,11 And
the longer children nursed, the better. Children who breastfed over 6 months
had lower chance of overjet, and they demonstrated wider inter- canine and
inter- molar width.12 A number of studies also found that extended
breastfeeding continued to decrease the risk of malocclusion, and the longer a
child breastfed, the less likely they were to have malocclusion.13,14
2
| IMPACT
OF AIRWAY OBSTRUCTION
A very young infant will typically
breathe quietly with lips closed. But even in early infancy, there are a number
of factors that can interrupt this process and change the course of
craniofacial growth. Airway obstruction has many aetiologies and is not
uncommon in early childhood.
Allergic rhinitis, with and without oral habits, has
been implicated in both anterior and posterior open bites.15 The
condition commonly known as ‘long adenoid face’ is marked by enlarged tonsils
or adenoids that accompany a retrognathic jaw, SMA and with larger lower
anterior face height.16 Otitis media is correlated with HNP and PCB.17
Septal deviation can result in a HNP, demonstrating the inter- relationship of
these facial features.18
3
| IMPACT
OF SOFT TISSUE RESTRICTION
Research on ankyloglossia diagnosis
and its impact on oral function is growing because of its implication in OSA.4
The frenulum's upper and lower insertion points, its thickness and flexibility
and length of free anterior tongue all impact lingual range of motion and oral
function. Ankyloglossia has been correlated with reduced inter- canine and
inter- molar width, SMA, HNP, AOB, overjet and incisor spacing.19
The vertical lift to the hard palate, and not horizontal extension past the
incisors, is the most accurate measure of normal lingual function.20
The existence of labial frenula is not a symptom of
dysmorphology, but variations in the insertion points, thickness and its
contribution to trapping liquid and food can have a negative impact on oral and
dental development. Maxillary labial ties can make breastfeeding difficult and
painful for the mother when the upper lip does not create enough of a flange to
adequately draw in more of the nipple.21 As teeth develop, central
incisors may separate, rotate or flare in response to a low fibrous frenulum.22
Buccal ties are the least researched of the oral frenula and their impact on
gingival recession and maxillary growth are not well documented.23
Pronounced buccal frenula contribute to pocketing of food in the vestibules.
4
| IMPACT
OF MOUTH BREATHING
If the impact of airway obstruction,
soft tissue enlargement and/ or soft tissue restriction is great enough, nasal
breathing may not be adequate for muscular and cognitive functions, and a
pattern of mouth breathing can develop. The sinuses experience their largest
growth in early childhood, and nasal breathing activates growth in occipital
and nasal joints and sutures of the facial bones.24 Mouth breathing
encourages a lower jaw posture which can change directional growth over time.
When compared to those with nasal breathing patterns,
mouth breathing was more highly correlated to HNP, PCB and AOB.25
Mouth breathing during the critical facial growth period was associated with a
‘clockwise’ rotation of the mandible and an increase in lower anterior face
height.5
Mouth breathing not only changes the anterior of the
face, but also changes the shape of the oropharyngeal airway. With an increase
in anterior face height, there is often a decrease in posterior height. Mouth
breathing has been associated with smaller retropalatal and retroglossal areas,
and lengthening of the pharynx, a risk factor for OSA.26
D'ONOFRIO | 45
Orofacial myofunctional disorder should be viewed on
a continuum, and as oral dysfunction influences the growth of oral structures,
the structure impacts oral function in return. Mouth breathing at night
contributes to other symptoms of SDB, including snoring. Children who snore are
more likely to have HNP and PCB.27 Mouth breathing at night, without
any other symptoms, ‘is a risk factor for OSA, and is associated with increased
disease severity and upper airway collapsibility.’28
Once a child has been diagnosed with OSA, they often
present with extreme malocclusions and dysmorphology. ‘Children with diagnosed
OSA had a significantly increased overjet, a reduced overbite, narrower upper
and shorter lower dental arches when compared with the controls. Snoring
children had similar but not as significant differences as OSA children when
compared to controls. There were more children with an AOB in the OSA group and
with a Class II or asymmetric molar relationship in the groups of OSA and
snoring subjects compared with non- obstructed controls.’29 This
study asserts that structural changes are caused by long- term functional
changes in the head, neck and tongue in order to maintain a patent airway
during sleep.29
Breastfeeding reduces the chances of a child developing
SDB. A study of school- aged children found those who were breastfed for only a
few months had less incidence of snoring and OSA than those who were bottle-
fed.30
5
| IMPACT
OF ORAL RESTING POSTURES
‘The ability to breathe effortlessly
and quietly through the nose with the tongue suctioned up and the lips gently
closed is essential to optimal craniofacial growth and development.’31
Muscular pressure on facial bones, or the lack thereof, can influence
directional growth over time. Open lip posture can encourage upper incisor
flaring.32 Lingual- palatal stability maintains the palatal arch and
supports the mid and lower anterior face. Low lingual resting posture has been
correlated with both Class II and Class III malocclusions.33 The
static lingual posture at rest slowly changes the face, the swallowing pattern
and occlusion.
6
| IMPACT
OF ORAL HABITS
Most dentists and orthodontists
understand that sucking and chewing habits contribute to AOB and PCB.15,34
However, instead of treating thumb sucking as an aberrant habit, evidence
indicates this behaviour may be a symptom of airway obstruction15
and/or ankyloglossia.35
In addition to inappropriate and uneven pressure into
the hard palate and alveolar process, oral habits contribute to keeping the
tongue low and forward in the mouth, which promotes an open mouth resting
posture and the cascade of effects that can follow.35 As expected,
the longer oral habits continue, the more severe the malocclusion.36
The occurrence of oral habits may be in response to
an appropriate biological need and breastfeeding may be the best prevention. An
inverse relationship was found between the duration of breastfeeding and the
occurrence of oral habits.37 Palatal stimulation appears to be a
necessary part of facial and cognitive development, and by allowing infants to
experience it through breastfeeding may not only promote better facial
development, but also reduce the likelihood of maladaptive oral habits.38
7
| IMPACT
OF SWALLOWING
Atypical swallowing develops as a
compensatory movement pattern when normal movement is inhibited in some way. A
tongue thrust swallow involves excessive perioral effort and the tongue exerts
forward and/or lateral pressure into the teeth, rather than vertical pressure
into the hard palate with a front to back motion.39 Lingual- palatal
stabilization for the swallow is far weaker in children with PCB.40
This swallowing pattern reinforces a low resting posture, contributing to HNP,
PCB and further malocclusion.41
8
| IMPACT
OF CHEWING
Chewing begins in the first year of
life and provides early sensory- motor awareness, oral proprioception and a
foundation for normal oral movement needed for speech. In addition to aiding in
the digestion of food, chewing stabilizes the temporomandibular joint42
and regulates bone growth.43 Chewing helps reduce psychological
stress,44 improve attention45 and increase cognition.46
Non- nutritive chewing of objects, imbalanced chewing
and inefficient chewing can contribute to the development of malocclusion.
Researchers are now speculating on how our diet has evolved over the centuries
and its possible contribution to craniofacial changes, specifically to
retrognathia.47 Softer foods require less chewing and less bite
force. Masseter orientation angle and bite force were found to be correlated to
different malocclusions, with Class III showing to have the greatest bite
force.48 Children with PCB demonstrated reduced bite force and
unbalanced jaw function.49
Because one symptom of OMD can contribute to the
development of another, it is logical to assume that mouth breathing and oral
habits can negatively impact masseter development. Compared to nasal breathing,
mouth breathing is shown to reduce the chewing stroke count and chewing cycles.50
Children with oral habits produced less bite force than children without such
habits.51
9
| IMPACT
OF OMD OVER TIME
As stated above, OMD is often the
result of a sequence of events or lack of intervention at critical periods. The
impact is cumulative. Children with low rates of breastfeeding, with oral
habits and mouth breathing during sleep present with more malocclusions.52
When OMD occurs during childhood, this disorder then becomes a contributing
factor in other diseases and disorders.28,53-55
The long- term concerns go well beyond poor facial
aesthetics. Unresolved OMD can contribute to serious dental and medical
conditions that threaten the quality and length of a person's life. If the jaw
is rotated back into the airway and the hard palate invades and deviates the
sinuses,56 it may be difficult to breathe nasally. As the face
grows, unbalanced pressure on the craniofacial bones can contribute to
temporomandibular dysfunction. Airway- based malocclusions and those correlated
to SDB are further complicated by co- occurring symptoms including clenching
and grinding, regarded together as bruxing. Bruxing can contribute to facial
pain and tooth damage.
New research into SDB and OSA links poor sleep and
airway obstruction to daytime behavioural disorders in children.53
The relationship between SDB and increased risk for academic and social failure
is also well documented.54 By the time a child reaches their teens,
their dysmorphic facial structure may put them at permanent risk for a lifetime
of airway function disorders.31 A study of facial measurements of
over 4000 teens concluded, ‘the combination of a long face, reduced nose
prominence and width, and a retrognathic mandible may be diagnostic facial
features of SDB that may warrant a referral to specialists for the evaluation
of other clinical symptoms of SDB.55
10
| IMPACT
OF MATERNAL ORAL DYSFUNCTION ON THE DEVELOPING FOETUS
Sleep- disordered breathing is common
during pregnancy and is linked to hypertension, gestational diabetes,
pre-eclampsia and foetal growth retardation.57 A retrospective study
of over 300,000 women at one military treatment facility found that women
diagnosed with OSA had higher rates of cesarean delivery, gestational hypertension,
pre- eclampsia and preterm delivery,58 and there were indications
that maternal OSA was a direct cause of foetal distress.59
Research suggests infants of mothers with OSA are
more likely to be born with a retrognathic jaw and HNP and premature infants
appear to be more prone to PCB.60 Those born small for gestational
age were noted to have a short anterior cranial base, increased lower anterior
face height, small retrognathic jaws with SMA and small maxilla. Interestingly,
this same study found dental age was not delayed, which extrapolates to normal
sized teeth in an underdeveloped face.61 Instead of starting out
with beautifully aligned facial morphology, these infants begin life at risk
for OMD and all the consequences that follow.
11
| CONCLUSION:
PREVENTION OF MALOCCLUSION
Orthodontists must advance their
thinking in how, when and why they are treating their patients. Closing an open
bite without resolving its underlying pathology increases the risk of
orthodontic relapse.62 The signs and symptoms of OMD can appear in
the first weeks of life but can also occur at any point in the lifespan.
In addition to providing structural
solutions to problems once they occur, dentists and orthodontists must play a
proactive role in preventing acquired craniofacial disorders and supporting
optimal craniofacial growth.
In response to a growing body of scientific and
clinical evidence, all medical and dental professionals have a responsibility
to screen for daytime and nocturnal breathing disorders, for enlarged and
restricted oral tissue in patients of all ages, and for feeding and oral
dysfunction early in life. Beautiful babies were meant to grow up to be
beautiful adults. Ira L Shapira DDS, DABDSM, DAAPM, FICCMO Neuromuscular Dentistry remains an enigma to many dentists who do not understand the purposes of the electrodes, the TENS, the computers and more. It has unfairly become a target of poor and misleading definitions by doctors who do not understand its basic principles. There are several basic premises that underlie Neuromuscular Dentistry. The first premise is that the stomatognathic muscles are the primary determinate of the mandibles position during all jaw functions (when the teeth are not in occlusion) and that rest position is one of the most important positions in dentistry. Rest position is a maxillary to mandibular jaw relation where the teeth are not in occlusion but are prepared to occlude. In Neuromuscular Dentistry Rest is a position of bilaterally equal and low muscle tonicity from which the mandible moves into full occlusion with minimal muscle accommodation. Following closure from rest position the mandible should return to rest with similarly balanced low muscle tonicity. Rest position is determined not only by the mandible’s relation to the cranium but also by the position of the head relative to the body, the suprahyoid and infrahyoid muscles and the position of the hyoid bone. To fully understand the relation of jaw movement to head posture read the Quadrant Theorem of GUZAY (Available from the ADA Library). In essence it shows in engineering terms that after accounting for both rotation and translation of the mandible the actual axis of rotation of the mandible is at the odontoid process of the second vertebrae not at the mandible condylar head.The second premise is that occlusion is important in neuromuscular dentistry as a resetting mechanism of the trigeminal nervous system’s control of the stomatognathic muscles. Myocentric occlusion is ideally a position in which the muscles move the mandible from a non-torqued rest position into full occlusion with minimal muscle accommodation and no interferences of occlusal contacts until full closure is attained thus eliminating all torque during closure. This means that there are no noxious contacts received by the periodontal ligaments or the muscular proprioceptors that must be avoided by the muscles (accommodation) but rather allow “free” entry into myocentric occlusion. The jaw muscles will return to rest position after closure with the muscles maintaining their healthy low tonicity. Relaxed healthy musculature is the gold standard of neuromuscular dentistry. Swallowing is a primary activity when the jaw is closed into full occlusion. In order to swallow it is necessary to fixate the mandible and this happens as the teeth occlude. During chewing, speaking and other jaw functions the teeth do not actually occlude in normal function but are separated (during chewing by a bolus of food). Typically swallowing occurs approximately 2000 times a day and is momentary accounting for 6-10 minutes maximum time in occlusion over the course of the day and acts as a neuromuscular reset switch for trigeminally innervated muscles. During a healthy swallow the teeth will move freely without interference into full occlusion with bilateral equal contact and bilateral equal muscle activity and then return to rest position with low muscle tonicity. A deviate swallow as evidenced by scalloping of the tongue is a sign of a possible TMJ disorder and is also 80% predictive of sleep apnea (80% predictive in Dental study- 70% predictive ENT study) Neuromuscular occlusion (myocentric) occurs when centric occlusion (maximum non-torqued intercuspation of teeth) is coincidental with a balanced muscle closure where the muscles will return to their relaxed state following closure. Myocentric is the ideal position for swallowing.The dentist utilizing neuromuscular techniques does not determine a specific position of the condyles in the fossa. The position of the disk and condyle are determined primarily by the teeth (bite or orthotic during occlusal correction therapy) in occlusion (myocentric) and the application of muscle activity. Neuromuscular dentistry allows the patients healthy relaxed muscles to determine the joint relations with the teeth serving as a neuromuscular reset switch during closure. Neuromuscular dentistry rejects the notion that manipulation of the patient’s jaw by the intervention of the clinician muscles are more important in determining the relation of the components of the TM Joint than the muscles of the patient. Centric relation is not used as a reference position for mounting casts on an articulator. Centric Relation is considered a border movement of the mandible and as reported in orthopedic literature joints are rarely used in their border positions. The HIP plane (defined by hamular notches, incisive papilla and the occipital condyles) and/or Campers plane is used to relate the maxilla to an articulator. When cosmetic considerations are involved photos are used to to incorporate this physiologic plane to soft tissues of the face. Ideally the occlusal plane (parallel to the HIP Plane) will bisect the odontoid process of the axis of the atlantoaxial joint the actual center of rotation for the mandible after accounting for translation and rotation according to the quadrant theorem. This occlusal plane will be at a 90-degree angle to gravitational force when the head is in an upright position. A neuromuscular bite is used to mount the mandibular casts according to data from EMG recordings and jaw tracings with TENS. The incorporation of the Curve of Spee based on Centro Masticale (CM) point which continues thru the mandibular condyle and the Curve of Wilson also based on CM point that is involved is stimulation of the autonomic nervous system via tongue reflexes when the lateral border of the tongue touches the lingual surfaces of the teeth. (Critical Reviews in Oral Biology & Medicine, Vol. 13, No. 5, 409-425 (2002)) This excellent article is available online at http://cro.sagepub.com/cgi/content/full/13/5/409 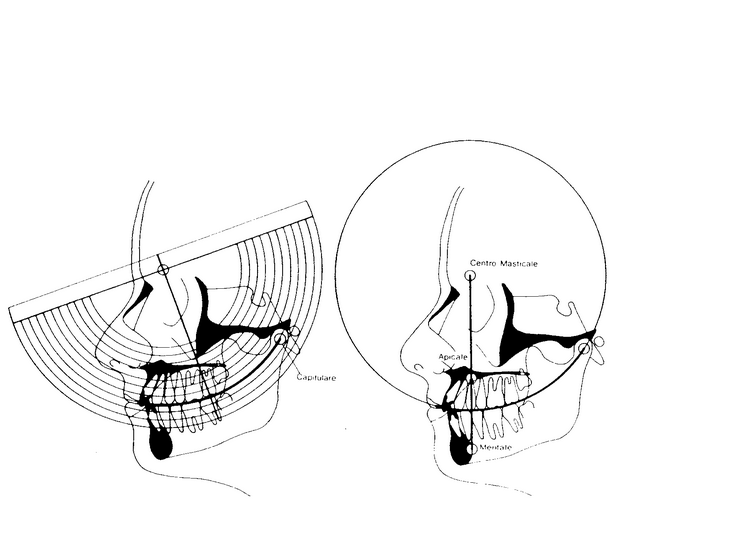 Neuromuscular dentistry considers many of the problems associated with TMD to be repetitive strain injuries. Movements become harmful when closure requires excessive accommodation and then the system fails at its weakest link. If the weakest link is muscle health we will see formation of tender sore muscles with eventual formation of taut bands and trigger points. If there is muscle overuse from clenching and/or grinding there will be post exercise pain secondary to anaerobic lactic acid build-up. If the weakest point is in the TM Joint then when repetitive strain occurs the muscle accommodation will lead to increased intra-articular pressure. This again will break down the joint at its weakest point. This may occur as a displaced disk or as wear of articular surfaces or many other conditions. It may come at the expense of the bone of the condyle leading to flattening or beaking of the condyle. Clenching and bruxism are two particularly well-known and harmful parafunctional habits that lead to varied repetitive strain injuries. There are many other parafunctions that can lead to problems. Some parafunctions are actually protective muscle accommodation such as the deviated or reversed swallow that protects the TM Joints and masticatory muscles at the expense of altered head, hyoid and spine position and consequent muscle problems. The second type of problems would be described as I/O or Input/ output errors in computer lingo. The CNS is essentially a biological computer and is affected by input from afferent nerves from the body. Autonomic system function include the fight or flight response with concomitant release of adrenaline that alters the heart rate, blood pressure, muscle tone etc. This system effects the Hypothalamus pituitary complex with feedback to the adrenals and effects on ACTH and cortisol levels. During periods of acute stress these effects have a positive survival value but during chronic stress become a liability to the individual as described by Hans Selye in his book “The Stress of Life” and his discussion of the General Adaptation Syndrome. There are numerous biochemical changes that occur in the brain secondary to aberrant or nociceptive input into the brain these can be affected by correction of the neurological input, by using drugs to change the brain chemistry or a combination of these approaches. Ideally correction of the underlying cause of these biochemical changes is the preferred method of treatment.  The utilization of TENS or transcutaneous neural stimulation over the coronoid notch has been shown by Mitani and Fujii (1974 J. Dent Res.) to block the motor division of the trigeminal nerve and relax the musculature via anti-dromic impulses (hyperpolarisation) to both the alpha and gamma motor neurons without influence from proprioceptive and nocioceptive (tooth contacts) inputs The TENS is then use to create a balanced synchronize pulsing to find the trajectory of closure where a myocentric registration can be obtained. The muscles will return to their relaxed position following closure into myocentric. The use of Computerized mandibular jaw tracking allows the dentist to measure and record resting jaw position relative to the cranium at a given head position. The location of myocentric occlusion is determined by the dentist with the aid of information from tracings recorded. Electromyography or EMG is used to record relative values of resting muscle activity of masticatory muscles as well as muscles such as Sternocleidomastoid or Trapezius. While there are no absolute “normal values” of resting muscles the clinician uses his information to compare muscle activity within a given patient. Muscles should be approximately equal activity bilaterally. Thomas 1990 in Frontiers of Oral Physiology vol 7 pp162-170 demonstrated that Spectral analysis of the post TENS EMG may be utilized to evaluate muscle fatigue and differentiate between muscle atrophy or fatigue or relaxed muscle states. This was later confirmed by Frucht, Jonas and Kappert at Frieberg University in 1995 (Fortschr.Kieferorthop vol 56 pp 245-253). Utilizing the two modalities together allows the clinician to evaluate the rest position of the jaw and simultaneously the health and functional activity of the muscles. The EMG also allows tests to evaluate the functional capacity of the muscles and again compare the right and left sides for symmetry. The use of functional recordings (during clenching and closure into centric occlusion) allows the clinician to evaluate whether or not muscle function is satisfactory and functional. The use of EMG also allows evaluation and correction of first contact of closure position within microseconds bases on firing order of the masseter and temporalis muscles. The first point of contact on closure is vitally important and equilibration of orthotics and dentition must be finely adjusted until first contact is evenly dispersed on posterior dentition. Neuromuscular dentistry is very concerned with the effects of mandibular position on the body as a whole and on the effects of the body on jaw and head position. The work of Sherrington and the righting reflex explains how ascending and descending disorders affect a patient. These phenomena have been best explained by Norman Thomas BDS, PhD. I will not attempt to explain this complicated topic in this agenda, which may be found in Anthology of ICCMO vol V pp159-170. It obviously must incorporate the Quadrant Theorem of Casey Guzay, the physiological aspects of the balance organ of the inner ear and the vestibular apparatus located in the brainstem as well as visual feedback. The control of sympathetic and parasympathetic systems by the cerebellum is quite intricate and also affected by head position. Correction of the chewing cycle is an important part of occlusal finalization. It must be understood that the chewing cycle is different on the each side and that interferences can occur on both the opening and closing strokes of the chewing cycle. Interferences in chewing strokes are easiest to detect by study of the chewing strokes on computerized mandibular scans (MKG). Head position during chewing is not is normally in the upright head position but in the feeding position approximately a 30-degree anterior head flexion. Correction of the chewing cycle is at least as important as correction of right, left and protrusive excursions. Chewing is a healthy function of the craniomandibular apparatus where as excursive movements are actually exercises in parafunctional movements.There is more commonality to treatment of TMJ disorders by neuromuscular and non-neuromuscular dentists than differences and complications caused by neural intensification in the reticular activating system, emotional aspects and the relation to the limbic system, connections to the sympathetic and parasympathetic nervous systems via the Sphenopalatine Ganglion and sympathetic chain, and chemical changes and cerebroplastic changes that occur during chronic pain leading to hyperalgesia and allodynia all can be discussed in greater detail and explained in relation to neuromuscular dentistry. The basics physiology including effects of Golgi tendon organs and muscle spindles on jaw muscles remain constant and must always be carefully considered during treatment. Barney Jankelson’s famous quote, “if it is measured it is a fact otherwise it is an opinion “ rings as true today as when he first said it. Neuromuscular dentistry is about making accurate measurements and the use of those measurements to improve the doctor’s ability to make a differential diagnosis and tailor treatment to relieve pain and create stable restorative dentistry with healthy relaxed musculature. I would like to make a disclaimer that this is my personal definition of Neuromuscular dentistry from 30 years of practice and to thank my mentors Barney Jankelson, Barry Cooper, Dayton Krajiec, Richard Coy, Harold Gelb, Peter Neff, Robert Jankelson and especially Jim Garry who first made me understand the connections between increased upper airway resistance and the common developmental aspects of sleep apnea and craniomandibular disorders. A special thank you for Dr Norman Thomas for his extraordinary help in understanding the complex physiology and anatomy underlying neuromuscular dentistry and in reviewing this paper prior to presentation. My personal research in the 1980’s as a visiting assistant professor at Rush Medical School examined the jaw relations of patients with obstructive sleep apnea based on neuromuscular evaluations of jaw relations with a Myotronic's kinesiograph and a myomonitor to find neuromuscular rest position. These studies showed jaw relations in the male apnea patients that were strikingly similar to those found in female TMD patients. The National Heart Lung and Blood Institute considers sleep apnea to be a TMJ disorder. The NHLBI published a report, “Cardiovascular and Sleep Related Consequences of TMJ Disorders” in 2001 that can be found at http://www.nhlbi.nih.gov/meetings/workshops/tmj_wksp.pdf Dr Shapira returned to Rush as an assistant professor at the sleep center in the 1990’s where he treated a wide variety of obstructive apnea patients with commercial and customized intraoral sleep appliances. He was a founding and credentialed member of the Sleep Disorder Dental Society that has become the American Academy of Dental Sleep Medicine, He is a Diplomate of the American Board of Dental Sleep Medicine, on the board of the Illinois Sleep Society, a Diplomat of the American Academy of Pain Management, a Regent Fellow of the International College of Cranio-Manibular Orthopedics and a representative of that group to the TMD Alliance. He is a long time member of AES, AACFP, Academy of Sleep Medicine and the Chicago Dental Society. Dr Shapira teaches hands-on in-depth Dental Sleep Medicine courses to small groups at his Gurnee office. A family history of genetic cancer led Dr Shapira into research on stem cells an he also holds several patents (method and device) on the collection of stem cells during early minimally invasive removal of the uncalcified tooth bud of developing third molars. This procedure can be complete in minutes with greatly reduced morbidity compared to current surgical techniques used for removal of developed third molars. He hopes in the future that patients will routinely remove the tooth buds and collect and save the stem cells for anti-aging and regenerative medical uses. |
Thoughts on Our Orthodontic Profession Fabio Savastano* International College of Neuromuscular Orthodontics and Gnathology, Italy Citation: Fabio Savastano. “Thoughts on Our Orthodontic Profession”. EC Dental Science 19.10 (2020): 18-19. *Corresponding Author: Fabio Savastano, International College of Neuromuscular Orthodontics and Gnathology, Italy. Received: May 28, 2020; Published: September 10, 2020 30 years have passed since I started my journey in Orthodontics, 25 of which working alone in my office. My wife and my daughter (yes, they are dentist too) share with me my fears and struggles now, comforting me with their presence and energy. Like every other passioned orthodontist, I’m still eager to test my knowledge and update on new trends and issues. I’m constantly in touch with my colleagues and other young dentists that in one way or another have been at my courses or are part of the master’s program where I teach. I gather opinions and watch out to be updated on new tendencies. I got to see TAD’s coming, NiTi and oral scanners just to name a few. I lived through several treatment modalities with aligners and miracle treatments on a weekly basis. Some have disappeared and probably we will never be remembered. Some could very well last and become part of our profession. Defining what a profession is, is not an easy task. It’s a job of course, a craft also. The expertise within should be the result of several components: an orthodontic professional is most certainly driven from a good shovel of vocational beliefs as well as love for the profession itself. A pro should look like a pro, talk like a pro and be wise like a pro. Is that it? Probably not. There is something missing here, and I will try to tell you what it’s about. Let’s take a look at what’s going on in orthodontics in general. First of all, I would like to express my sincere disgust on a new trend that’s taking place all over the world: the superfast treatment 3-3, upper lower or both. Are we really going in the direction of a new sort of “professional” figure, the cosmetic orthodontist? Second, again I would like to express my sincere doubts on the new trend "sagittal first" for the treatment of Class II. This concept represents the milestone of ignorance in orthodontic diagnosis and treatment that crushes crowds of young orthodontists and leads them classify a Class II only as a sagittal problem. Class II is and remains a vertical problem. There is something going on here that is well beyond professionalism. The people that guide all this cannot be so ignorant, I will not accept that, but I can accept that they have lost the most important thing a great leader should have, honesty. They know exactly what they are doing, they are feeding that hungry crowd of what they want most, fast easy and money-making results. Who cares if several patients develop TMD, malocclusion has nothing to do with TMD! (again, I’m disgusted). My last words must focus on something positive, especially after this darn Covid thing. There are orthodontists out there who are leaders in the field and really do their best to guide and change the world. It’s like I’m saying there are some good guys out there that do merit continuous ovations for their effort that appears genuine and separated from the trends I just talked about. You don’t really have to believe in them 100% and embrace their philosophy and interpretation of orthodontics, but I assure you that if you keep your perspective wide and take a look at what they are saying (shouting sometimes) you will be somewhat surprised. I’m going to give you a list of suggested readings and I certainly know that the choice is yours. I know that you will read the list for sure, and I know that some of you will be curious enough to buy a book or click on a website. My greatest effort here is not this suggested reading stuff (even though I wish you would read it), it’s a more deepened message that I hope you get: beware of detractors. Never in these 30 years have I seen such disregard for the Orthodontic Profession. Never in these 30 years I have felt the attack coming from orthodontic companies like in these days. They promote their products through illegitimate orthodontists that have sold their souls for money and promote their pseudoscience with purchased publications. Suggested Readings 1) Jaws, the story of a hidden epidemic, by Sandra Kahn and Paul R. Ehrlich. 2) Buteyko: Close Your Mouth: Buteyko Clinic Handbook for Perfect Health, by Patrick McKeown and Paul Metcalfe. 3) And don’t forget this website on Orthotropics of my friend Mike Mew: https://orthotropics.com/orthotropics-thinkingbeyond- teeth/ |
Neuromuscular Orthodontic Correction of a Class III Patient Fabio Savastano* and Giulia Savastano Abstract Introduction: Patients with a skeletal Class III pattern without reverse overjet may show signs and symptoms of temporomandibular joint (TMJ) overload. Orthodontic correction should be performed while paying particular attention to TMJ preservation. Mandibular tracking and surface electromyography (SEMG) are essential aids for diagnostic and follow-up procedures. Aim: To apply neuromuscular dentistry principles to the orthodontic treatment of skeletal Class III patients with signs and symptoms of TMJ disfunction. Methods: The diagnostic and follow-up procedures were based on the use of mandibular tracking (computerized mandibular scanning), SEMG and transcutaneous electrical nerve stimulation. Results: After treatment and at 10.5 years follow-up, there were no TMD signs or symptoms. Conclusion: Neuromuscular orthodontics is an effective diagnostic procedure that can assist the orthodontist in correcting malocclusions related to TMJ disorders. Keywords: Neuromuscular Orthodontics; Transcutaneous Electrical Nerve Stimulator; Mandibular Posture; Mandibular Kinesiograph; Surface Electromyography |
| Neuromuscular dentistry and the role of the autonomic nervous system: Sphenopalatine ganglion blocks and neuromodulation. An International College of Cranio Mandibular Orthopedics (ICCMO) position paper Ira Shapira, USA The Sphenopalatine Ganglion (SPG) is known to play an integral role in the pathophysiology of a wide variety of orofacial pains involving the jaws, sinuses, eyes and the trigeminal autonomic cephalalgias. It supplies direct parasympathetic innervation to the trigeminal and facial nerves. Sympathetic innervation from the superior sympathetic chain passes thru the SPG to the trigeminal and facial nerves.This paper reviews relevant and significant literature on SPG Blocks and Neuromodulation published in peer reviewed medical and dental journals. Neuromuscular Dentistry employs ULF-TENS to relax musculature and simultaneously provide neuromodulation to the ganglion.Conclusion: The effects of ULF-TENS on the autonomic nervous system acts on the Limbic System and Hypothalamus (H-P-A) to address Axis II issues during neuromuscular dental procedures. It also directly affects the autonomic component of the trigeminal nerve involved in almost all headaches and migraines as well as the Myofascial and Joint disorders of TMD. Shapira, Ira. (2019). Neuromuscular dentistry and the role of the autonomic nervous system: Sphenopalatine ganglion blocks and neuromodulation. An International College of Cranio Mandibular Orthopedics (ICCMO) position paper. Cranio: the journal of craniomandibular practice. 37. 201-206. 10.1080/08869634.2019.1592807. |
Neuromuscular Dentistry: Use and Abuse Savastano, Fabio ** International College of Neuromuscular Orthodontics and Gnathology (ICNOG), Albenga, Italy Full article at https://www.ecronicon.com/ecde/pdf/ECDE-18-00951.pdf |
| Applying neuromuscular techniques in the orthodontic setting. Savastano, Fabio * * International College of Neuromuscular Orthodontics and Gnathology (ICNOG), Albenga, Italy in oral function, hold promise for the treatment of a spectrum of oral disorders. Indeed, such technologies will help patients receive a more targeted level of care. Here we present a case report concerning an 11-year-old boy who underwent orthodontic treatment for recurrent pain of the right temporomandibular joint (TMJ), misaligned teeth, and irregular clicking noises (associated with the right TMJ) during mouth opening. The basic principles of the use of mandibular tracking, surface electromyography, and transcutaneous electrical nerve stimulation (TENS) to diagnose malocclusions and determine the cranio-mandibular relationship are outlined. Case presentation: Pre-treatment status, progress, post-treatment status and 8-year follow-up data are shown. Conclusion: As neuromuscular orthodontics can provide detailed functional analyses through a combination of technologies, the clinician is better placed to evaluate the needs of the patient and deliver treatment. Further deployment of such techniques should, therefore, be encouraged to increase orthodontic health and practice. http://aseestant.ceon.rs/index.php/sejodr/article/download/15529/5731 |
| Effects of upper cervical manipulation on the electromyographic activity of the masticatory muscles and the opening range of motion of the mouth in women with temporomandibular disorder: randomized and blind clinical trial Efeitos da manipulação cervical alta sobre a atividade eletromiográfica dos músculos mastigatórios e amplitude de movimento de abertura da boca em mulheres com disfunção temporomandibular: ensaio clínico randomizado e cego Los efectos de la manipulación cervical en la actividad electromiográfica de los músculos masticatorios y la amplitud del movimiento de apertura de la boca en mujeres con trastorno temporomandibular: un ensayo clínico aleatorizado y ciego Gustavo Luiz Bortolazzo1, Paulo Fernandes Pires2, Almir Vieira Dibai-Filho3, Kelly Cristina dos Santos Berni1, Bruno Mascella Rodrigues4, Delaine Rodrigues-Bigaton5 Mailing address: Delaine Rodrigues Bigaton. Avenida Jaime Pereira, 3701 – CEP: 13403-900 – Piracicaba (SP), Brazil. E-mail: drodrigues@unimep.br – Phone: (19) 97872013 – Presentation: Oct. 2015 – Accepted for publication: Dec. 2015 – Approved by the Ethics Research Committee of the Universidade Metodista de Piracicaba (UNIMEP), under protocol number 01/09 and registered in the Brazilian registry of clinical trials (RBR-4j6xfx). DOI: 10.590/1809-2950/15568322042015 Study performed in the Laboratory of Therapeutic Resources – School of Health Sciences of the Universidade Metodista de Piracicaba (UNIMEP) – Piracicaba (SP), Brazil. 1PhD, Graduate Program in Oral and Dental Biology, Universidade de Campinas (UNICAMP) – Piracicaba (SP), Brazil. 2PhD student, Graduate Program in Human Movement Sciences, Universidade Metodista de Piracicaba (UNIMEP) – Piracicaba (SP), Brazil. 3PhD student, Graduate Program in Rehabilitation and Functional Performance, Universidade de São Paulo (USP) – Ribeirão Preto (SP), Brazil. 4Undergraduate in Physiotherapy (IC), Universidade Metodista de Piracicaba (UNIMEP) – Piracicaba (SP), Brazil. 5Professor, PhD by the Graduate Program in Human Movement Sciences, Universidade Metodista de Piracicaba (UNIMEP) – Piracicaba (SP), Brazil. ABSTRACT | We evaluated the effects of upper cervical manipulation on the surface electromyographic activity (sEMG) of masticatory muscles and range of motion of the opening movement of the mouth in women with temporomandibular disorders (TMD). We evaluated 10 women with myogenic a TMD diagnosis, according to the Research Diagnostic Criteria for Temporomandibular Disorders (RDC/TMD) and divided randomly into an experimental group (EG) n=5, which received upper cervical manipulation, and a placebo group (PG) n=5, which received maneuvers without therapeutic effects. Five interventions were performed in both groups, once a week, with performance of pre-intervention assessments, post-immediate assessments (after 1st intervention) and post-delayed assessments (48 hours after the 5th intervention). The sEMG activity was processed using the root mean square and normalized by the peak value (RMS EMGn). We used for comparison the Student’s 426 t-test and ANOVA two-way repeated measures, adopting as significance the amount of 5%, and the Cohen d for treatment effect size. We found a significant interaction of group vs time (p<0.05) in the RMS EMGn of the left and right temporal muscles at rest, as well as for all masticatory muscles during maximal isometric contraction during jaw elevation and jaw-depression. Treatment effect size, high to moderate, was observed in the EG, especially in the post-delayed assessment. We also observed a significant increase (p<0.05) and a high treatment effect during mouth opening in the EG. The upper cervical manipulation demonstrated a balance of the RMS EMGn of the masticatory muscles and increase the opening range of motion of the mouth in women with myogenic TMD. Keywords | Manipulation, Spinal; Electromyography; Range of Motion, Articular; Temporomandibular Joint Disorders. |
| Does orthodontic proclination of lower incisors in children and adolescents cause gingival recession? Sabine Ruf, DDS, DrMedDent,a Ken Hansen, DDS, OdontDr,b and Hans Pancherz, DDS, OdontDrc Giessen, Germany, and Malmo¨, Sweden In this investigation we sought to assess the effect of orthodontic proclination of lower incisors in children and adolescents with respect to the possible development of gingival recession. Ninety-eight children with a mean 6 SD start-of-treatment age of 12.8 6 1.4 years, treated with the Herbst appliance, were surveyed, for a total of 392 lower incisors. Lateral head films, dental casts and intraoral photographs were analyzed with respect to the degree of orthodontic proclination, crown height, and gingival recession. In all subjects, Herbst treatment resulted in varying degrees of lower-incisor proclination (mean 5 8.9°, range 5 0.5° to 19.5°). In 380 of the surveyed teeth (97%), either no recession developed or preexisting recession remained unchanged during Herbst therapy. In only 12 teeth (3%) did recession develop or preexisting recession deteriorate during treatment. No interrelation was found between the amount of incisor proclination and the development of gingival recession. In conclusion, orthodontic proclination of lower incisors in children and adolescents seems not to result in gingival recession. (Am J Orthod Dentofac Orthop 1998;114:100-6.) |
| The temporomandibular joint in juvenile idiopathic arthritis: frequently used and frequently arthritic. Sarah Ringold*1 and Randy Q Cron2 Address: 1Department of Pediatrics, Division of Rheumatology, Seattle Children's Hospital, Seattle WA, USA and 2Department of Pediatrics, Division of Rheumatology, University of Alabama at Birmingham, Birmingham AL, USA Email: Sarah Ringold* sarah.ringold@seattlechildrens.org; Randy Q Cron - rcron@peds.uab.edu Pediatric Rheumatology 2009, 7:11 Abstract Recent recognition of the markedly high prevalence of temporomandibular joint (TMJ) arthritis in children with juvenile idiopathic arthritis (JIA) coupled with the significant morbidity associated with TMJ damage has prompted increased interest in both the clinical and pathological aspects of TMJ arthritis. This review focuses on the prevalence of TMJ arthritis in JIA, the imaging modalities used to detect TMJ arthritis, and the treatment of TMJ arthritis in children with JIA. |
| Prevalence of clinical signs of intra-articular temporomandibular disorders in children and adolescents A systematic review and meta-analysis Cristhiani Giane da Silva; Camila Pachêco-Pereira, DDS; André Luís Porporatti, DDS, MSc; Maria Gorete Savi, MSc; Marco A. Peres, DDS, MSc, PhD; Carlos Flores-Mir, DDSc, MSc, DSc, RCDC(C); Graziela De Luca Canto, DDS, MSc, PhD Background. The aim of this systematic review and meta-analysis was to assess the prevalence of clinical signs of temporomandibular joint (TMJ) disorders in children and adolescents. Type of Studies Reviewed. The authors selected only studies in which the investigators’ primary objective was to evaluate the prevalence of signs of TMJ disorders according to the international Research Diagnostic Criteria for Temporomandibular Disorders (RDC/TMD) in children and adolescents. The authors performed electronic searches without language restriction in 5 databases. The authors also assessed quality. Results. In this review and meta-analysis, the authors included 11 articles that described studies in which 17,051 participants had been enrolled. The overall prevalence of clinical signs of intra-articular joint disorders was 16% (95% confidence interval [CI], 11.59-19.94; n ¼ 17,051). The prevalence of TMJ sounds (click and crepitation) was 14% (95% CI, 9.67-19.79; n ¼ 11,316). The most prevalent sign was clicking (10.0%; 95% CI, 7.97-12.28; n ¼ 9,665) followed by jaw locking (2.3%; 95% CI, 0.56-5.22; n ¼ 5,735). Conclusions and Practical Implications. One in 6 children and adolescents have clinical signs of TMJ disorders. The results of this systematic research study can alert dentists about the importance of looking for signs of TMD in children and adolescents. Key Words. Evidence-based dentistry; prevalence; children; adolescents; temporomandibular joint disorders. JADA 2015:-(-):--- http://dx.doi.org/10.1016/j.adaj.2015.07.017 |
The role of biochemistry and neurophysiology in the re- education of deglutition Fellus Patrick* Qualified specialist, Former Consultant for the Hospitals of Paris, France Medical and Clinical Archives Review Article ISSN: 2515-1053 Abstract The transition from suction-type deglutition to dentition-type deglutition should spontaneously occur between ages 3 and 4, but the genetic information is not always systematically expressed. The new program must then be engrammed by cortical or sub-cortical pathways. Introduction Suction-type deglutition, the physiological mode for swallowing saliva in the young infant, is a praxis developed in the brainstem in utero. It will remain physiological as long as the primary dentition is not established and mastication has not begun. But beginning at age 4, a new deglutition program is naturally implemented via a subcortical pathway in 60% of children: dentition- type deglutition. By the muscular force it generates, it promotes optimal growth of the maxillae [1]. But endogenous or innate factors can only provide the potential. “They will only be expressed if adequate exogenous conditions occur in a timely manner” (JP Changeux) [2,3]. Dawson Church states that “genes are activated or inactivated by our beliefs, emotions and attitudes.” There are two principal causes for the persistence of suction-type deglutition: ––the child has never had the opportunity to discover this new function ––the child has discovered it, but the limbic system, the gateway to engramming a new program, has not retained it, usually for psychological reasons (immaturity, thumb, pacifier, bottle, food too soft). Most children requiring orthodontic treatment are found in the latter category. The re-education of orofacial functions is consequently a necessity accepted by nearly all practitioners during orthodontic treatment, but it is necessary to properly understand both neuroanatomy and physiology to achieve reproducible and controllable results. Deglutition re-education What strategy to implement? - hope that the normalization of dental arches will lead to a change in function. This is more feasible when wearing a removable retainer. The introduction of an element modifying proprioceptive sensations will automatically lead to a change in the afferent message, and consequently the efferent message [4]. The anatomical part having been modified by treatment, the correct program can install itself naturally, but without control, the new praxis may also remain dysfunctional. However, care must be taken with bonded retainers. If the functional modification occurs, there are no negative consequences, but if the dysfunction remains, the pathological muscular force will be iatrogenic, no longer in the dental alignment but in the supporting bone tissue, which may be the source of subsequent periodontal disease. -use of functional devices, usually at night, lingual envelope-type (NLE) devices intended to modify the lingual posture, Robin device, lingual elevator, Farrell or Bergensen-type gutters. However, the results remain haphazard, as long as the dysfunctional commands are not inhibited, aside from the fact that wearing these devices may be difficult for the child to deal with and rejected by the limbic system. -consultprofessionals,suchasspeechtherapistsorphysiotherapists. The child must initially become aware of what he/she is doing, then the maneuver he/she should be doing, and its repetition should enable automation. Eric Kandel, Nobel Prize in Medicine recipient in 2000 for his work on the transition from short-term to long-term memory, demonstrated inthesecasesanincreaseintheactivityofneurotransmittersinthesynapses involved, but we remain in the domain of short-term memory [5,6]. “Memory is not based on the properties of nerve cells as such, but on the nature of the connections between neurons and how they process the sensory information received”: learning consists of tracing new circuits, and this plasticity occurs either by reconfiguring existing programs or creating new ones (Figure 1 and Table 1). Correspondence to: Fellus Patrick, Qualified specialist, Former Consultant for the Hospitals of Paris, France, E-mail: fellusp@wanadoo.fr Key words: deglutition, short-term memory, procedural memory Received: June 09, 2017; Accepted: June 21, 2017; Published: June 24, 2017 Med Clin Arch, 2017 doi: 10.15761/MCA.1000106 Volume 1(1): 1-3 Patrick F (2017) The role of biochemistry and neurophysiology in the re-education of deglutition Table 1. Reconfiguring existing circuits. 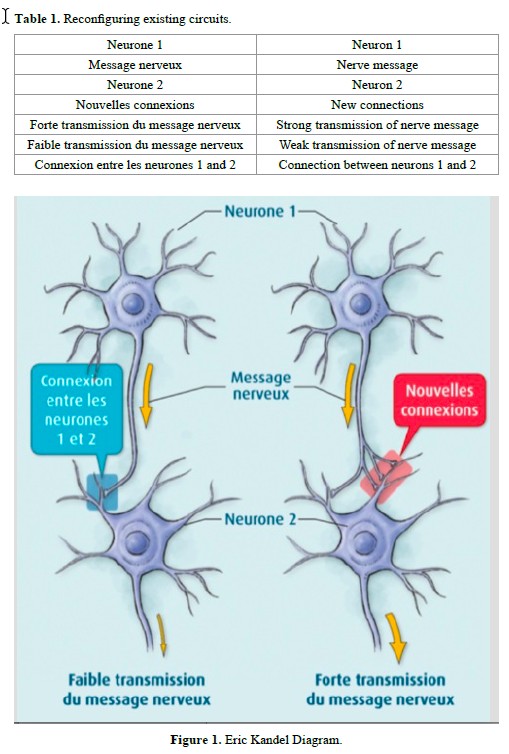 Understanding the transition from short-term memory to long- term memory was clarified by the work of Kandell in Aplysia, by reconstituting a simplified neural circuit: a single sensory neuron connected to a single motor neuron: -A slight stimulus releases neurotransmitters at the synapse, but the nucleus is not involved; this is short-term memory (weekly speech therapy sessions). This information will remain available only briefly. -If the stimuli are repeated soon after, a dialogue occurs between the synapse and the nucleus to activate CREB* and produce a new protein, essential for the transition to long-term memory. This new CPEB** protein present in the synapse will function as a prion and permanently ensure the transmission of the message. *CREB (Cyclic AMP Response Element-Binding Protein), a protein that activates the genes responsible for long-term memory. CREB 1 is the activator and CREB is the 2 inhibitor. **CPEB: (Cytoplasmic Polyadenylation Element-Binding Protein), a transcriptional regulatory protein in the synapse which contributes to the stabilization of long-term memory. Creating new circuits A highly emotional state can short-circuit normal constraints and produce a sufficient quantity of MAP-kinase* molecules, which will be sent to the nucleus to inactivate CREB-2** molecules and facilitate CREB-1** activation and direct imprinting of this experience in long- term memory *MAP-kinase kinase: acts in conjunction with protein kinase A, to initiate long-term memorization. - FroggyMouth is a device worn a rather short time, 15 minutes a day while watching television (a reward recognized by the limbic system), which will force the child to discover a new deglutition method via the sub-cortical pathway; thus, not by stimulating neurotransmitter activity, but by creating new synapses. In fact, by no longer being able to close the lips, the child will be unable to swallow by suction, aspirating between the anterior and posterior mouth, triggering an abrupt and immediate reaction in the brainstem: find a new deglutition program. The simultaneous contraction of the levator muscles of the mandible in a stable and comfortable dental occlusion with those of the soft palate and styloglossus will allow a peristaltic movement of the tongue (provided that the anatomical environment is compatible) and disconnect the tongue-lips synkinesis. This new deglutition program will be immediately integrated into long-term memory by the creation of a new neural circuit. But this is only the first step, which is necessary but not sufficient for the transition to automation. Automation The child will then have two programs for swallowing saliva, and just as on a computer when there are two programs, it is the activation of one or another icon that initiates its execution (Figures 2 and 3). The therapist should therefore monitor the resting posture for relaxation of the perioral muscles and dental occlusion upon swallowing. Control by the trigeminal nerve, solicited at this step, will replace control by the facial nerve and inhibit the role of the latter. This necessary inhibition of the faulty circuit is fundamental to automation of the correct program. Only FroggyMouth allows this double action. The trigeminal nerve, which also controls the respiration centers in the pontine tegmentum through its sensory nucleus, will promote restoration of nasal respiration, allowing the tongue to adopt a high posture posteriorly (the lingual dome). “This dual posterior and occlusal functional necessity of the tongue, too often forgotten by re-educators of orofacial function, is likely one of the causes of the too-frequent failures of re-education” (Delaire) [7]. Similarly, contraction of the tensor tympani muscle, innervated by the trigeminal nerve, will allow ventilation of the middle ear by dilating the Eustachian tube, decreasing serous otitis problems Control may be transferred to the parents, who need to observe lip posture 5 times/ day, and who then congratulate or correct the child (Figures 4 to 8). These two actions are not similar, involving cortico-cortical circuits that traverse the basal ganglia and cortico-cortical circuits that traverse the cerebellum [8]. Wearing FroggyMouth starting at age 3 has no contraindications. Conclusion Early normalization of orofacial functions, regardless of the technique chosen, will enable a three-step preventive approach: Med Clin Arch, 2017 doi: 10.15761/MCA.1000106 Volume 1(1): 2-3 Patrick F (2017) The role of biochemistry and neurophysiology in the re-education of deglutition  Figure 2. The suction-type deglutition icon is activated by the facial nerve: “My lips are contracted, my teeth do not touch.” Figure 3. The dentition-type deglutition icon: “my lips are relaxed, my molars are in occlusion” is activated by the trigeminal nerve, whichallows not onlymolar occlusion but also the protection of the tongue from biting, due to the abundance of trigeminal nerve endings in its epitheliallining. Figure 4. Contrary to what speech therapists suggest, attention needs to be paid not to the tip of the tongue, but rather to its posterior portion. Obsessedwith the sensorysearch for the retro-incisive papilla, the child risks raising the tip of the tongue, leading to lowering of the posterior portion, which will prevent the involvement of the styloglossus, the levator muscle of the lingual dome. Figure 5. Worn for 15 min daily while watching television, FroggyMouth allows relaxation of all anterior facial muscles. Figure 6. Spontaneous improvement of an incisor gap after wearing Froggy Mouth for one month. Figure 7. Resumption of treatment with only Froggy Mouth after repetition of a previous orthodontic treatment. Figure 8. Case treated by Gérard Altounian. -prevent deformities from appearing, -if they occur, prevent them from worsening, References 1.Patrick F (2003) Orthodontie précoce en denture temporaire Cdp. 2.Jean-Pierre C (1983) L’homme neuronal Fayard. 3.Gérard C (2010) Les oralités humaines Doin. 4.Gérard C (2015) Oralité du fœtus Sauramps Médical. 5.Arthur G (1996) Neurosciences. Piccin. 6.Eric K (2011) A la recherche de la mémoire. Odile Jacob. 7.Patrick F, Waddah S, Lalauze-Pol R (2016) De la dysfonction à la dysmorphose. Apport de Froggy mouth. Edition Orthopolis. 8.Fournier M, Girard M (2013) Acquisition and sustainment of automatic reflexes in maxillofacial rehabilitation. Orthod Fr 84: 287-294. [Crossref] Copyright: ©2017 Patrick F. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Med Clin Arch, 2017 doi: 10.15761/MCA.1000106 Volume 1(1): 3-3 |
MASTICATORY FUNCTION IN TEMPOROMANDIBULAR DYSFUNCTION PATIENTS: ELECTROMYOGRAPHIC EVALUATION FUNÇÃO MASTIGATÓRIA EM PACIENTES COM DISFUNÇÃO TEMPOROMANDIBULAR: AVALIAÇAO ELETROMIOGRÁFICA Giédre BERRETIN-FELIX1, Katia Flores GENARO2, Inge Elly Kiemle TRINDADE3, Alceu Sergio TRINDADE JÚNIOR3 1- DDS, MSc, PhD, Professor, Department of Speech, Language and Audiology, Bauru Dental School, University of Sao Paulo (FOB-USP), Bauru, SP, Brazil. 2- DDS, MSc, PhD, Associate Professor, Department of Speech, Language and Audiology, Bauru Dental School, University of Sao Paulo (FOB-USP), Bauru, SP, Brazil. 3- DDS, MSc, PhD, Associate Professor, Department of Biological Sciences, Bauru Dental School; University of Sao Paulo (FOB-USP), Bauru, SP, Brazil. Corresponding address: Department of Speech Pathology and Audiology, University of São Paulo - Al. Dr. Octávio P. Brisolla, 9-75, 17012- 901 - Bauru - SP - Brazil. - E-mail: gfelix@fob.usp.br Received: February 25, 2005 - Modification: May 10, 2005 - Accepted: June 30, 2005- J Appl Oral Sci. 2005;13(4):360-5 Temporomandibular dysfunction (TMD) is a complex disturbance that involves the masticatory muscles and/or temporomandibular joint, causing damage to the masticatory function. This study evaluated the electromyographic activity of the masseter muscle during habitual mastication of bread, apple, banana, cashew nut and paraffin film (Parafilm M) in 25 adult subjects, of both gender, with TMD. The results were compared to those of a control group, composed of 15 adult subjects, of both sexes, free of signs and/or symptoms of TMD. The MYO-TRONICS Inc., K6-I computer software was used for electromyographic processing and analyzed the following parameters: duration of the act, duration of the masticatory cycle and number of cycles. No significant differences were found between subjects in the control group and individuals with TMD as to duration of the masticatory act and of the masticatory cycle, considering all materials used for mastication. The duration of the masticatory act and cycle was longer during mastication of paraffin film in both groups. The number of masticatory cycles was higher for mastication of apple in comparison to mastication of banana, in both groups. It can be concluded that the consistency of foods influences the duration parameters of the act, duration of the cycle and the number of masticatory cycles, and the behavior of the masticatory muscles in individuals with TMD during habitual mastication is similar to that verified in individuals without TMD. Uniterms: Mastication; Electromyography; Food; Temporomandibular joint disorders. |
